Faculty A-Z

Daniel MacPhee
Associate Professor of Reproductive/Developmental Biology B.Sc.(Hons.) PEI, Ph.D. Western OntarioRetired BioMedical Sciences
t: 709-777-8543; f: 709-777-7010
Email:
dmacphee@mun.ca
Address:
Division of BioMedical Sciences Rm 5335, Health Sciences Centre Memorial University of Newfoundland St. John's, Newfoundland and Labrador A1B 3V6
The Role of Focal Adhesion Signalling in Uterine Smooth Muscle During Pregnancy
Overview: The onset of labour is normally under precise temporal control, so as to ensure the timely delivery of a term fetus whose organ systems are sufficiently mature for survival in the extrauterine environment. Signals that initiate labour are known to reside in the fetal genome. These signals ultimately lead to a switch in the phenotype of the uterine smooth muscle, the myometrium, from a muscle which during pregnancy is quiescent to one which is spontaneously active, excitable, highly responsive to uterine agonists, and exhibits a high degree of cell-cell coupling. Today, it is beginning to be appreciated that growth and cytoskeletal remodelling of myometrial cells during late pregnancy may also be critical for myometrial function during labour and delivery.
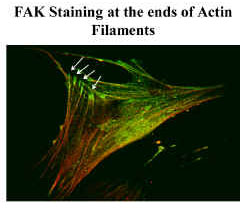
In smooth muscle, the transmission of force between the contractile apparatus of the cell and the extracellular matrix is thought to occur at membrane-associated dense plaques or focal adhesions. Focal adhesions consist of clusters of transmembrane receptors named integrins, heterodimeric proteins that consist of α and β monomers, that mediate interactions between the extra- and intracellular environments. The cytoplasmic regions of integrins connect with cytoskeletal elements and signalling components such as Focal Adhesion Kinase (FAK), while the extracellular regions connect to specific extracellular matrix molecules such as collagen, laminin or fibronectin.
During contraction, smooth muscle exhibits a plasticity of its mechanical response that is not well accounted for on the basis of the traditional sliding filament paradigm. There is growing evidence that actin polymerisation and the dynamic remodelling of the actin cytoskeleton play key roles in the regulation of smooth muscle contraction. FAK has been implicated in signalling pathways regulating cytoskeletal organisation, cell motility and contraction in other cell types, but direct evidence supporting a role for FAK and other focal adhesion-associated proteins as regulators of actin dynamics in differentiated smooth muscle tissues, including the myometrium, is currently lacking.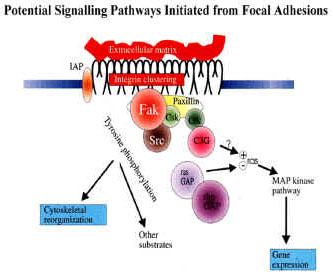
Goals: The rat myometrium is an excellent model tissue for studying the basic cellular mechanisms of smooth muscle contraction. It is easily obtainable from pregnant rats and this facilitates study of the role of focal adhesion signalling in contraction during late pregnancy and during labour. My research goal, in the short term, is to identify which integrins and focal adhesion-associated proteins are the major players in myometrial function and contractile activation during late pregnancy and labour.
Research and Socio-Economic Significance: My research will greatly enhance our understanding of the basic cellular mechanisms of smooth muscle contraction. Our research also has socio-economic significance. In about 5-10 % of pregnancies in North America, the onset of labour occurs pre-term. This figure may even be higher in particular population groups and has not fallen over the past 20-30 years. Pre-term birth is associated with a 70% rate of neonatal death and up to 75% of neonatal morbidity. In addition, the cost of caring for pre-term babies in the USA alone has been estimated at $8 billion annually. The underlying cause(s) for the majority of pre-term births still remains a mystery because scientists and clinicians do not fully understand the underlying biochemistry and molecular biology of pregnancy and parturition. This research project will provide novel information on the biochemical and molecular mechanisms of uterine myometrial function during pregnancy and labour. In the future, this information will greatly facilitate the development of effective therapeutic strategies to prevent pre-term labour.
Funded by NSERC
The Role of Integrin-Linked Kinase in Human Trophoblast Differentiation
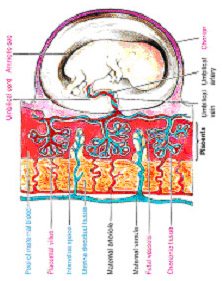
The physiological relationship between the mother and fetus is mediated by the placenta. The placenta is crucial for nutrient and gas exchange that ultimately controls the growth and viability of the fetus and, as a result, the health of the mother. Unfortunately, the current understanding of human placental development is insufficient to allow determination of the origin and subsequent resolution of conditions or diseases such as unexplained miscarriages and preeclampsia.
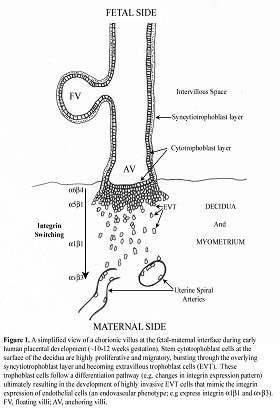
In the placenta, physiological exchange of gas and nutrients occurs via chorionic villi consisting of outer syncytiotrophoblast cells, underlying cytotrophoblast cells, and a stroma. Floating villi represent the majority of villi and are bathed in maternal blood aiding gas and nutrient exchange. During early placentation (early 1st trimester), polarized cytotrophoblast cells in floating villi proliferate and differentiate, fusing to form the syncytiotrophoblast layer. The syncytiotrophoblast layer is a bi-directional interface for nutrient and gas exchange in the intervillous space but it is also a critical contributor to the endocrine environment of pregnancy. Alternatively, polarized cytotrophoblast cells migrate through the syncytium and form columns of non-polarized extra-villous trophoblast (EVT) cells that connect the embryo to the uterine wall. Proximal EVT cells of the columns are proliferative while more distal EVT cells become highly invasive invading the decidua, spiral arterioles (displacing vascular smooth muscle cells and replacing endothelial cells), and the inner third of the uterine muscle layers. These latter processes of EVT differentiation are likely responsible for the relatively undisturbed and increased maternal blood volume available for physiological exchange in the intervillous space.
A cytoplasmic, focal adhesion-associated, serine/threonine kinase named integrin-linked kinase (ILK) is known to be a regulator of cell-ECM and cell-cell adhesion. Our hypothesis is that integrin-linked kinase regulates the differentiation of trophoblast cells during placental development. We believe that an understanding of this differentiation process may lead to a better understanding of the etiologic bases of placental disorders and/or diseases during pregnancy. The aims of this research project are 1) To identify where and when ILK is present in human placenta during gestation, 2) To evaluate whether ILK is a key enzyme for trophoblast differentiation.
 |
 |
Funded by CIHR/IRIF
What are the responsibilities of being a scientist? Check out the following video.
Graduate Students in the Lab:
Bryan White (PhD Candidate)
Trina Butler (PhD Candidate)
Pia Elustondo (PhD Candidate)
Mandy Peach (MSc Candidate)
Recent Publications:
White, BG, Williams, SJ, Highmore, K, MacPhee, DJ. 2005. Small heat shock protein 27 (Hsp27) is highly induced in rat myometrium during late pregnancy and labour. Reproduction 129: 115-126. [pubmed link]
Keywords: stress proteins, uterus, pregnancy, labour, myometrium.
1 Joint first authorship.