Southern Blot analysis of DNA

Southern Blot analysis of DNA
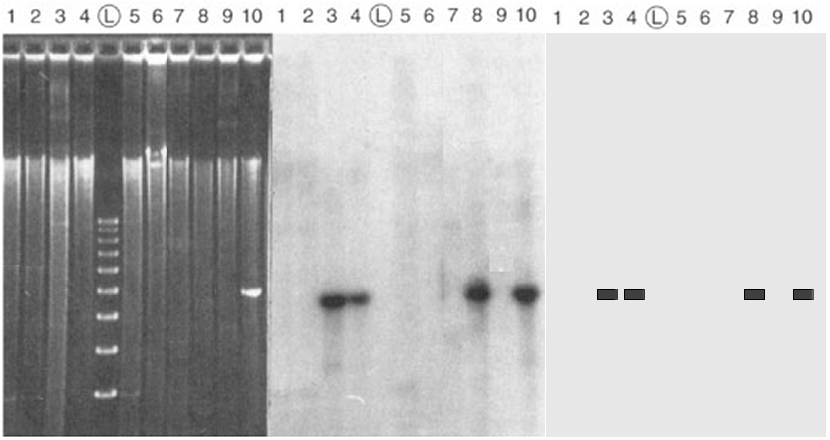
The left panel is an electrophoretic gel stained with ethidium bromide. Total DNA has been extracted from nine bacterial DNA clones (lanes ##1-9), digested with a particular restriction endonuclease, and separated by electrophoresis. Starting about one-third of the way from the top, a bright smear is present in each lane, indicating the presence of a random collection of restriction fragments of various sizes. There are no discrete bands, as no one DNA sequence is present in more than one copy in a haploid bacterium. The lane marked (L) is a "Ladder", a molecular weight standard with a series of DNA fragments at 100 bp intervals. A cloned DNA fragment has been loaded in lane #10: its mobility corresponds to the fourth rung of the ladder, about 400 bp.
The middle panel
is a Southern Blot autoradiogram of
the same gel. The DNA in the gel is transferred ("blotted")
to a nitrocellulose filter. The filter is then hybridized with a
radioactively-labelled DNA (a "probe")
made from the same DNA loaded
in lane #10. Where the probe DNA finds a
complementary sequence in the blot, it base-pairs ("sticks")
to that DNA. The filter is then overlaid with a piece of X-ray
film: the probe exposes the film at that point, and produces
a dark band.
The right
panel is a schematic representation of the autoradiogram. The
information content is the presence or absence of bands, and the size of the fragments, in each lane. The
autoradiogram show that a DNA sequence homologous to the probe DNA is present in clones ## 3, 4, & 8,
with the expected size as indicated in lane #10.
The DNA is absent in clones ##1, 2, 5, & 9.
The probe sticks to itself in control lane #10, as
expected. The analysis shows that the gene of interest has been
successfully cloned in the first set, which can now be analyzed
further.
Figure modified from © 2000 by Griffiths et al.; text material © 2026 by Steven M. Carr