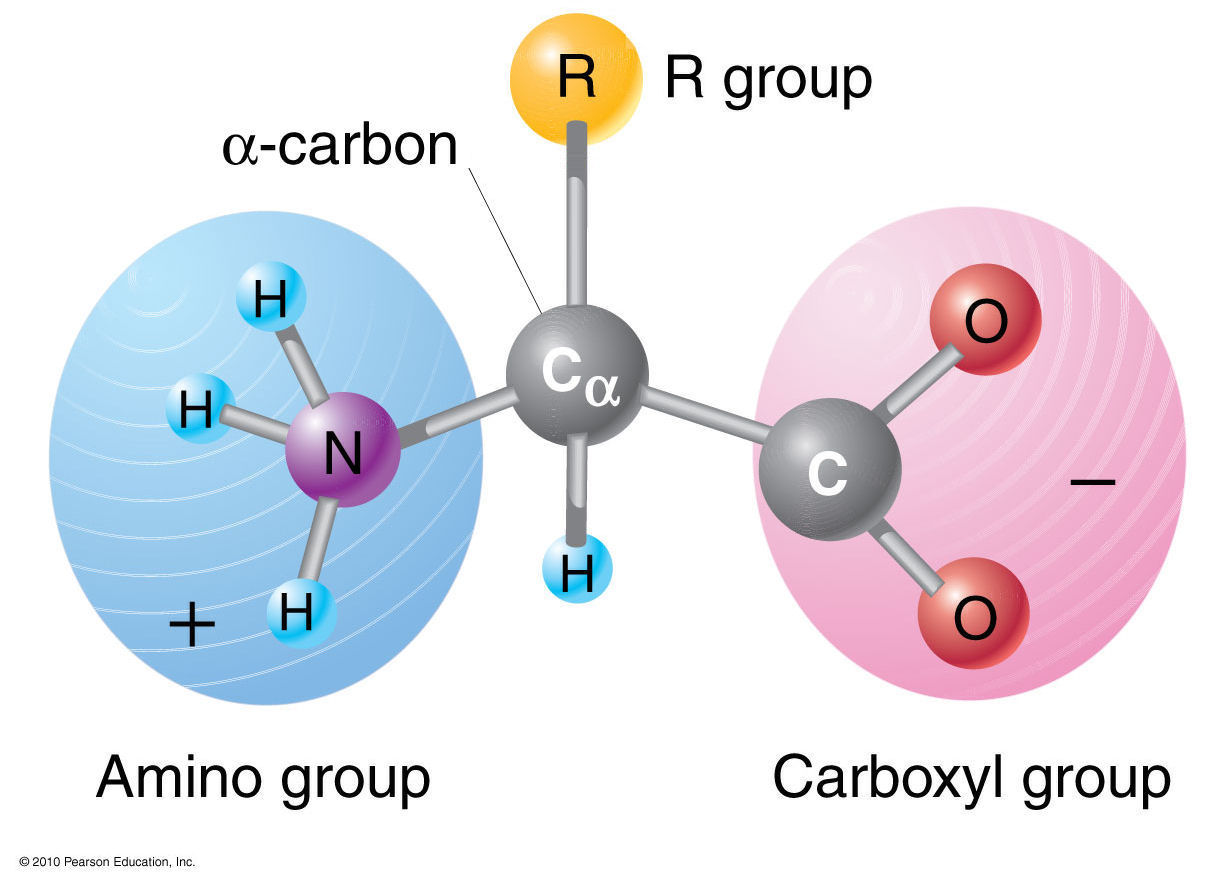
Stereoisometry of Amino Acids
The four bonds
of the central or alpha
carbon (C![]() ) of an amino
acid are directed towards the four corners of a tetrahedron. With respect
to the carboxyl (COO-)
and amino (NH3+)groups, there are two
possible arrangements of the H and Radical group. These
arrangement are literally mirror images of each other, and are
called stereoisomers (AKA
enantiomers).
Stereoisomers are designated D (dextro-rotatory)
or L (levo-rotatory) according
to the direction in which the crystalline forms rotate
polarized light, to the right and left, respectively.
Naturally-occuring amino acids are exclusively of the L form
) of an amino
acid are directed towards the four corners of a tetrahedron. With respect
to the carboxyl (COO-)
and amino (NH3+)groups, there are two
possible arrangements of the H and Radical group. These
arrangement are literally mirror images of each other, and are
called stereoisomers (AKA
enantiomers).
Stereoisomers are designated D (dextro-rotatory)
or L (levo-rotatory) according
to the direction in which the crystalline forms rotate
polarized light, to the right and left, respectively.
Naturally-occuring amino acids are exclusively of the L form