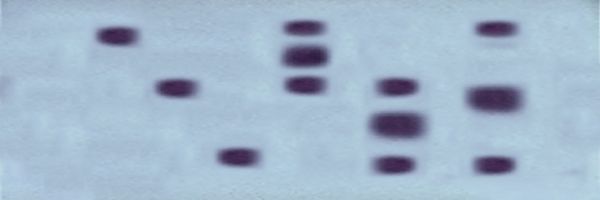
Use of protein electrophoresis to detect allozyme variation
Non-synonymous DNA mutations
result in the replacement of one amino acid by another with a
different electrical charge. This results in a slight
modification of the net charge of the protein. These protein
variants are called allozymes, because they are encoded by
different alleles at an enzyme gene
locus. [Allozymes should not be confused with isozymes,
which are different forms of the same enzyme
encoded at different gene loci].
Allozyme
variation is detected by means of protein electrophoresis.
Tissue extracts are placed in a solid support medium (a gel, typically starch or
cellulose acetate) in slots (wells)at one end, called the
origin. An electrical
field is applied with the negative (cathodal) end at the
origin, and the positive (anodal) end opposite. Because
most proteins have a net negative charge, they migrate
away from the cathodal origin
towards the anode at the other end of the
field. [Positively charged protein move towards the cathode].
The rate of migration depends on the ratio of charge
to mass of the protein. After several hours, the positions
of the allozymes may be detected either directly by staining, or
by a coupled enzymatic reaction that links the enzyme substrate
to a colored dye. The result is an electropherogram.
IMPORTANT: Be sure to
distinguish between the observed allozyme phenotypes
on the gel, and the inferred allele genotypes
in the DNA.
In the examples
below, the cathodal
end is at the top and the anodal end at the bottom.
In the first example, an analysis of the monomeric (single
subunit) enzyme Alcohol Dehydrogenase (ADH)
identifies three bands in lanes 1, 2, & 3 with slow, medium, or fast mobilities (measured
from the cathodal end). These
phenotypic enzyme patterns indicate that
the individuals have homozygous genotypes for
one of three alleles, which we call s, m,
or f, respectively. These genotypes may
be designated ADHss,
ADHmm, and ADHff,
respectively. The next three individuals show the patterns
expected from the three possible heterozygous genotypes, each of which has two bands:
ADHsm,
ADHsf, and ADHmf,
in lanes 4, 5, & 6, respectively.

In the second example, analysis of the dimeric
(two subunit) allozyme Transcendentalase (TRN)
identifies three bands in lanes 1, 2, & 3 with slow, medium,
or fast mobilities,
corresponding to homozygotes
of the alleles s, m, or
f : TRNss,
TRNmm, and
TRNff,
respectively. As before, the two alleles in each of the
heterozygous genotypes encode alternative protein subunits
with different electrophoretic charges. However, in
forming the dimeric protein, these subunits
combine as three products in the ratio 1 : 2 :1
. The heterozygous TRNsm,
TRNsf, and
TRNmf
therefore produce three-banded phenotypes,
recognizable by the mobilities of the products of the
homozygotes, with an intermediate band with twice the
protein product in the heterozygote.