Non-Standard base
pairing of rare tautomers
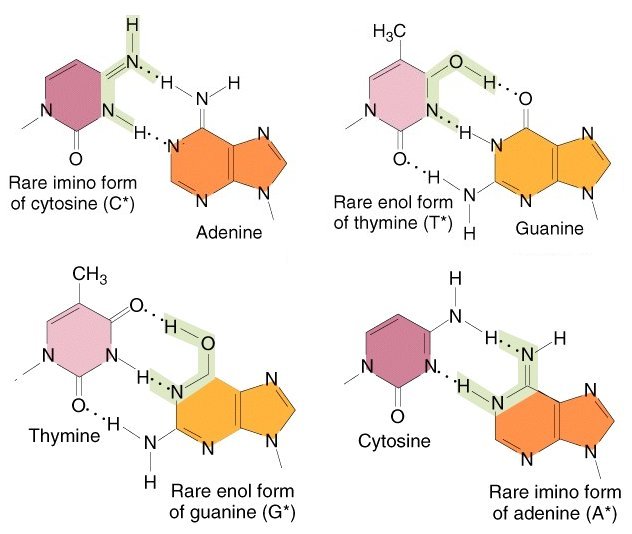
Specificity of base pairing between two-ring purines and one-ring pyrimidines is determined by the number of hydrogen (H) bonds available. Recall that the standard forms of C and G pair with three H-bonds, and the standard forms of A and T pair with two.
The rare tautomeric form C* has only two H-bonds, and pairs with the alternative purine, A. Likewise, the rare tautomeric form T* has three H-bonds, and pairs with the alternative pyrimidine, G.
In contrast, the rare tautomeric form G* retains the correct number of H-bonds, but the tautomeric arrangement of bonds forces these three bonds on standard T. The rare tautomeric form A* retains two bonds but forces them on standard C.
The net result is that the rare tautomers pair with the alternative ("wrong") purine or pyrimidine: C* with A, T* with G, G* with T, and A* with C, respectively.
Figure ©2000 by Griffiths et al.; text ©2026 by Steven M. Carr