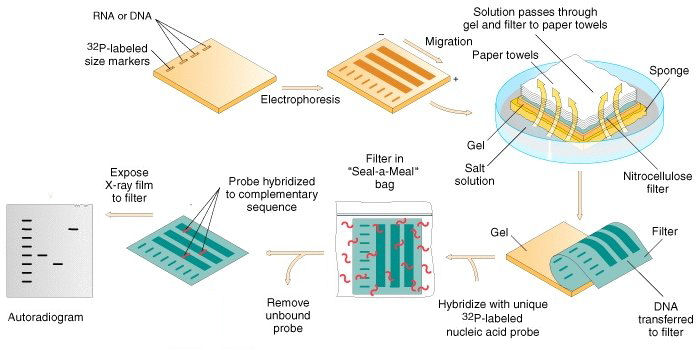
Analysis of DNA by the Southern Blot technique

Analysis of DNA by the Southern Blot technique
DNA is
applied to an agarose gel, and electrophoresis
separates the fragments of DNA according to size. The
gel is then placed atop a thin sponge
wick resting in a dish of salt solution, and a
nitrocellulose filter is placed on top of the
gel. A stack of absorbent material (paper towels,
borrowed from the washroom) is placed on top of this stack. The
absorbent material draws the salt solution from the dish into
the wick and through the gel by capillary action. The DNA
fragments are transferred to the nitrocellulose fibers.
The procedure is called a "Southern transfer" or
"blot" after the scientist Eric Southern who
invented the procedure. The filter now contains the
DNA fragments in the same pattern as the gel.
The filter is placed in a standard "Seal-a-Meal" bag that contains a solution of radioactively-labelled DNA probe [~~~~~] complementary to the gene sequence of interest. The DNA is denatured to separate the strands, and the probe binds to the filter only where a complementary DNA sequence is located. After washing to remove unbound probe, a piece of X-ray film is placed over the hybridized filter and exposed for several hours to several days. The radioactive label produces a black band on the film where it has stuck to the complementary DNA, producing an autoradiogram. If a labelled size marker (a "ladder") has been used, the exact sizes of the fragments can be determined. In other applications, the simple presence of a band in some lanes but not others provides the necessary data.
The Southern Blot
technique is useful for identifying a DNA sequence that
appears only once or twice in the genome, the typical situation
with diploid nuclear loci. Note that in this example, the
electrophoresis gel shows a continuous "smear" of DNA
of all fragment sizes, whereas the autoradiogram identifies
exactly three fragments, of three different sizes.
NB:
"Southern Blot" separation of DNA was
named for Eric Southern. A similar process of separation and
detection of RNA fragments was dubbed a "Northern
Blot", on the notion that North & South
like RNA & DNA are "opposites." Not to be
outdone, "Western" and "Eastern"
blots were developed as methods of separating and detecting
Proteins, and the "Northwestern" blot combines
Northern and Western blots in the same experiment.