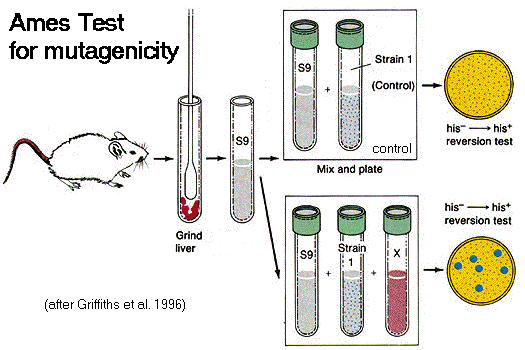
The Ames Test for
mammalian environmental mutagenicity
The Ames Test
combines a bacterial revertant
mutation assay with a simulation of mammalian metabolism in
vitro to produce a highly sensitive test for mutagenic
chemicals in the environment.
[Left] A rat liver homogenate is prepared to
produce a metabolically active extract (S9). [Top Right] The extract is combined with a
strain of his- Salmonella bacteria:
in the absence of histidine,
the bacteria are unable to grow on minimal medium (control result). [Bottom Right] The homogenate and
bacterial strain are combined with a suspected mutagenic substance
(X). The induction of revertant colonies indicates
that some his-
bacteria have mutated (reverted) to his+
, and therefore that substance X is a mutagen.
Different bacterial strains are sensitive to different types
of mutation.
Initial experiments used the reversion assay
without a liver homogenate. However, mutagenicity in
contrast to toxicity is not the result of
ingestion of a suspect substance, but rather its accumulation and
that of its breakdown products in the body. Use of a liver
homogenate simulates the metabolic breakdown of the suspected
mutagen in a mammalian system, and more accurately predicts
mutagenicity of substances ingested by humans. For example, sodium nitrate (NaNO3), which occurs naturally in smoked meats such as
bacon, hot dogs, ham, etc., is not itself mutagenic. However, when
acted upon by HCl in the
stomach, it is converted to nitrous
acid (HNO2), which has
been demonstrated to be a powerful mutagen by the Ames Test.
Bruce Ames (1928 - ) and his
undergraduate students tested large numbers of commercial products
in student labs at UC Berkeley when the test was first introduced
in the 1970s. Many common items, such as hairspray and food colours, were discovered to be mutagenic and were
withdrawn from the market. Ames also established that many
mutagenic compounds are also carcinogenic, an early
indication that changes in DNA sequence led to cancer.
Illustration
after
Griffiths et al. ©1996; All text material ©2026 by Steven M. Carr

